Crystallography and Crystal Chemistry Laboratory
- Home
- Instruments and Personnel
- Methods and Applications
- Project and Scientific Interests
- Pubblications
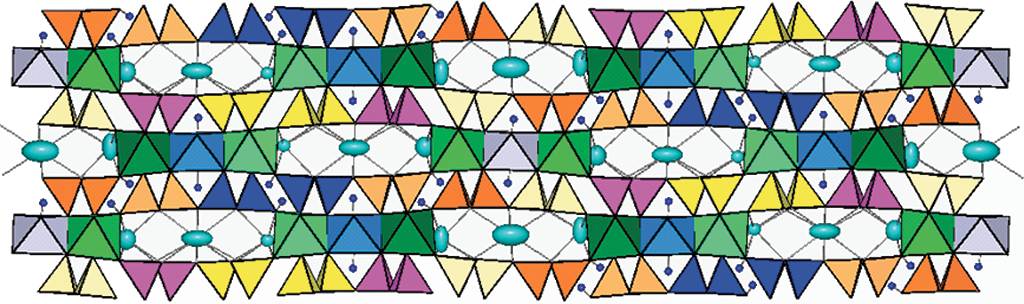
The worldwide community has recently celebrated with the International Year of Crystallography (IYCr2014, UN A/RES/66/284) the revolution brought in modern science by the discovery of X-ray diffraction and the development of crystallography. The capability of determining and visualizing the 3D structure of crystalline materials allowed scientists to successfully address very complex issues such as how structure may determine and tune properties, how structure and crystal chemistry depend on crystallization conditions, how structure is related to functional properties in geological, biological and technological processes.... Because scientists now have a detailed knowledge of most of the materials which play roles in our environment and even in our body, they can optimize their properties to better achieve present and future goals. This is true in chemistry, in pharmacology, in biology and in medicine, in Earth and materials sciences, in the conservation of our cultural heritage, etc.
The crystallography and crystal-chemistry lab in Pavia was founded in the ‘70s by the National Research Council with the name Centro di Studio per la Cristallografia Strutturale. For many years, the lab has provided data for many Italian groups working in Earth sciences. Many instruments were bought during the years, and they were often modified in house to improve specific capabilities of interest for Earth sciences. Our approach has always tried to integrate crystallography with geochemistry to provide detailed information of petrogenetic relevance. The chemical and structural information was conveyed into databases for specific groups of rock-forming minerals, which were then used to statistically model their mutual relations and improve our knowledge of how cation ordering is related to the conditions of crystallization. In the case of the amphiboles, the lab is still a reference for the international community. A further strength of the lab is the capability of monitoring in situ all those processes which occur at high T in crystalline materials, such as structure rearrangements, phase transitions, deprotonation processes.
All the research projects are addressed, in collaboration with scientists working in IGG or worldwide, in a strongly multidisciplinary way both as far methodologies (other diffraction- or spectroscopy-based techniques) and expertises (mineralogy, geochemistry, petrology) are concerned. Moreover, the lab provides companies or university labs with crystallographic data or structural models on a service basis..

INSTRUMENTS
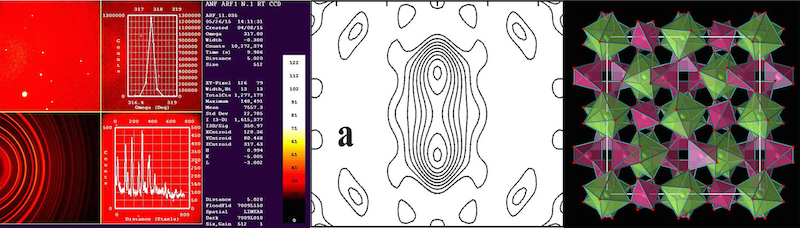
- One Bruker-AXS SMART-APEX single-crystal diffractometer (in partnership with Dipartimento di Scienze della Terra e dell'Ambiente - DISTA - of the Università degli Studi di Pavia), equipped with a CCD detector to allow for the fast collection of diffracted intensities at RT (Fig.1a). We use a 2 KW Mo X-ray tube.
- Two single-crystal four-circles diffractometers Philips PW1100 (Fig. 1b,c) run by the software FEBO (which has been written and developed in Pavia for a faster and more accurate analysis and collection of diffracted intensities). Again, they are run with a 2 KW Mo X-ray tube.
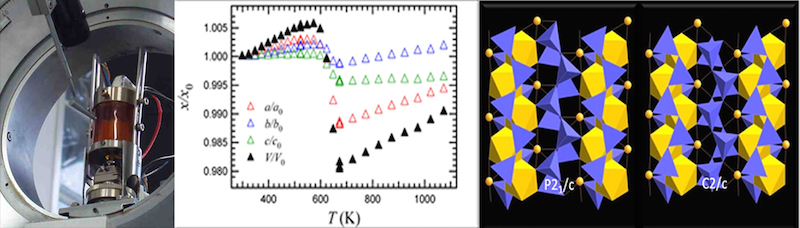
- A heating device (micro-furnace) designed and built in house, which can be mounted on the Philips goniometers (Fig. 2). It allows heating experiments and in situ measurements up to 1000 °C, with a theta range limited to 29°
- A powder diffractometer run by the DISTA is also available to identify mineral phases or organic and inorganic synthetic materials (also in mixtures).

STAFF
Dr. Roberta Oberti (CNR Researcher - Head Laboratory)
Roberto Gastoni (CNR Technician)
Other members of the team:
Giuseppe Toscani, (CNR Technician)
Dr. Serena Tarantino, DISTA Pavia (Associated Researcher)
Dr. Michele Zema, DISTA Pavia (Associated Researcher)
Massimo Boiocchi, Centro Grandi Strumenti, Pavia (Technician)
CONTACTS
Phone:
+39 0382 985885 (Dott.ssa Roberta Oberti)
E-mail:
METHODS
Whatever the specimen to analyse – a previously unknown mineral to characterize, a crystal extracted from a thin section because it is particularly useful to constrain a petrogenetic process, a new synthetic material of unknown structure and composition, the procedure is the same.

We select a crystal with the best optical behaviour (or a few crystals if we note some variability in colour) and mount it on the goniometer head.

We collect diffracted intensities, correct them for experimental parameters, treat the results taking into account the correct symmetry, solve the crystal structure, refine the crystal-chemical model which best reproduces atomic arrangement and site composition, check the electron density map to detect missing atoms, positional disorder or any feature that may improve the model, and eventually show up the crystal structure on our pc screen.
If the sample under investigation is a rock-forming mineral, we compare the results in the framework of our databases and improve the description in terms of composition and cation order, and then detect and quantify light and volatile elements based on the results of the structure refinement.
If needed, we use spectroscopic techniques (XAS, FTIR, FTIR imaging, Mössbauer) to double-check and improve information on oxidation states, local order, sample heterogeneity, chemical zoning or HT diffusion throughout the crystal.
In this way, crystal-structure refinement becomes a real chemical probe at the atomic scale.
APPLICATIONS
- Identification of crystalline phases (organic, inorganic, synthetic of found in Nature) via unit-cell parameters, powder diffractograms or complete structure refinement
- Structure identification and refinement at RT (theta max 67°)
- Mineral systematics and detailed crystal-chemical studies at RT
- Structure refinement at high T (up to 1000 °C, theta max = 29°)
- Thermal expansion, phase transitions, deprotonation processes monitored in operando in the same ranges of T and resolution.
Further expertise:
- Identification of different phases in mixtures and quality control by powder diffractometry
- Preparation of refined single-crystals for EMP and/or SIMS analysis (embedded in epoxy and brought to clean surfaces)
The staff is currently engaged in the characterization of new minerals and in completing the systematics of the amphibole supergroup (with particular attention paid to the oxo-component) and its modelling. Simple and accurate relations are being developed to allow calculation of crystal-chemical composition and cation order starting from the results of the structure refinement. They will be made available to the scientific community worldwide. Moreover, deprotonation processes in amphiboles and other hydrous minerals are currently studied by HT in situ experiments.
The staff is also involved in geochemical and petrological focused research projects, to which it provides information of the amount and partitioning of major and trace elements in minerals and on the OH content in amphiboles. All these features depend both on bulk and mineral chemical composition and on the P, T and fO2 conditions of crystallization.
This work can be done on minerals and material found in Nature or synthetized in the lab under controlled conditions.
The staff has long-lasting collaborations with many scientists in Italy and worldwide.
SELECTED PUBLICATIONS
- CÁMARA F., OBERTI R., CHOPIN C., MEDENBACH O. (2006) The arrojadite enigma: I. A new formula and a new model for the arrojadite structure. Am. Mineral. 91, 1249-1259.
- McCallum I.S., Domeneghetti M.C., Schwartz J.M., Mullen E.K., Zema M., CÁMARA F., McCammon C., Ganguly J. (2006) Cooling history of lunar Mg-suite gabbronorite 76255, troctolite 76535 and Stillwater pyroxenite SC-936: The record in exsolution and ordering in pyroxenes. Geochimica Cosmochimica Acta 70, 6068-6078.
- OBERTI R., .QUARTIERI S., DALCONI M.C., BOSCHERINI F., IEZZI G., BOIOCCHI M. (2006) Distinct local environments for Ca along the non-ideal pyrope-grossular solid-solution: a new model based on crystallographic and EXAFS analysis. Chem. Geol. 335, 347-359.
- Domeneghetti M.C., Fioretti A.M., CÁMARA F., Molin G., Tazzoli V. (2007) Thermal history of ALH 84001 meteorite by Fe2+-Mg ordering in orthopyroxene. Meteor. Planet. Sciences 42, 1703-1710.
- HAWTHORNE F.C., OBERTI R. (2007) Amphiboles: Crystal-chemistry. In: Amphiboles: Crystal Chemistry, Occurrence and Health Issues edito da F.C. Hawthorne, R. Oberti, G. Della Ventura e A. Mottana. RIM&G 67, 1-54.
- HAWTHORNE F.C., OBERTI R. (2007) Classification of the amphiboles. In: Amphiboles: Crystal Chemistry, Occurrence and Health Issues edito da F.C. Hawthorne, R. Oberti, G. Della Ventura e A. Mottana. RIM&G 67, 55-88.
- OBERTI R., DELLA VENTURA G., CÁMARA F. (2007) New amphibole compositions: natural and synthetic. In: Amphiboles: Crystal Chemistry, Occurrence and Health Issues edito da F.C. Hawthorne, R. Oberti, G. Della Ventura e A. Mottana. RIM&G 67, 89-124.
- OBERTI R., HAWTHORNE F.C., CANNILLO E., CÁMARA F. (2007) Long-range order in amphiboles. In: Amphiboles: Crystal Chemistry, Occurrence and Health Issues edito da F.C. Hawthorne, R. Oberti, G. Della Ventura e A. Mottana. RIM&G 67, 125-172.
- TIEPOLO M., OBERTI R., ZANETTI A., VANNUCCI R., FOLEY S. (2007) Trace-Element Partitioning Between Amphiboles and Silicate Melts. In: Amphiboles: Crystal Chemistry, Occurrence and Human Health edito da F.C. Hawthorne, R. Oberti, G. Della Ventura e A. Mottana. RIM&G 67, 417-452.
- WELCH M.D., CAMARA F., DELLA VENTURA G., IEZZI G. (2007) Non-ambient in situ studies of amphiboles. In: Amphiboles: Crystal Chemistry, Occurrence and Health Issues, edito da F.C. Hawthorne, R. Oberti, G. Della Ventura e A. Mottana. RIM&G, 67, pp. 223–260.
- CÁMARA F., OBERTI R., OTTOLINI L., DELLA VENTURA G., BELLATRECCIA F. (2008) The crystal-chemistry of Li in gadolinite: a multi-analytical approach. Am. Mineral. 93, 996-1004.
- CÁMARA F., NESTOLA F., ANGEL, R.J., OHASHI H. (2009): Spontaneous strain variations through the low temperature displacive phase transition of LiGaSi2O6 clinopyroxene. Eur. J. Mineral. 21, 599-614.
- Redhammer G.J., Cámara F., Alvaro M., Nestola F., Tippelt G., Prinz S., Simons J., Roth G., Amthauer G. (2010) Thermal expansion and high temperature P21/c-C2/c phase transition in clinopyroxene-type LiFeGe2O6 and comparison to NaFe(Si,Ge)2O6. Phys. Chem. Minerals, 37, 685-704.
- HAWTHORNE F.C., OBERTI R. (co-chairs), HARLOW G.E., MARESCH W.V., MARTIN R.F., SCHUMACHER J.C., WELCH M.D. (2012) Nomenclature of the amphibole supergroup. Am. Mineral. 97, 2031-2048.
- Nestola F., Pasqual D., Welch M.D., Oberti R. (2012) The effects of composition upon the high-pressure behaviour of amphiboles: compression of gedrite to 7 GPa and a comparison with anthophyllite and proto-amphibole. Min. Mag. 76, 987-995.
- PERINELLI C., ANDREOZZI G.B. CONTE A.M., OBERTI R., ARMIENTI P. (2012) Redox state of subcontinental lithospheric mantle and metasomatism relationships: insights from spinel-peridotites from northern Victoria Land (Antarctica). Contrib. Mineral. Petrol.164, 1053-1067.
- JENKINS D.M., DELLA VENTURA G., OBERTI R, BOZHILOV K. (2013) Synthesis and characterization of amphiboles along the tremolite-glaucophane join. Am. Mineral. 98, 588-600.
- OBERTI R., BOIOCCHI M., HAWTHORNE F.C., BALL, N.A., HARLOW, G.E. (2015) Eckermannite revised. The new holotype from the Jade Mine Tract, Myanmar: crystal structure, mineral data and hints on the reasons for the rarity of eckermannite. Am. Mineral. 100, 909-914.
- Dyar M.D., Breves E.A., Gunter M.E., Lanzirotti A., Tucker J.M., Carey C.J., Peel S., Brown E.B., OBERTI R., Lerotic M., Delaney J.S.(2016) Use of multivariate analysis for synchrotron micro-XANES analysis of iron valence state in amphiboles. Am. Mineral. 101, 1171-1189.
- OBERTI R., DELLA VENTURA G., BOIOCCHI M., ZANETTI A., HAWTHORNE F.C. (2016) New data on the crystal-chemistry of oxo-mangani-leakeite and mangano-mangani-ungarettiite from the Hoskins mine and their impossible solid-solution - An XRD and FTIR study. Min. Mag., in press.